Ciclo de Qualidade Analítica
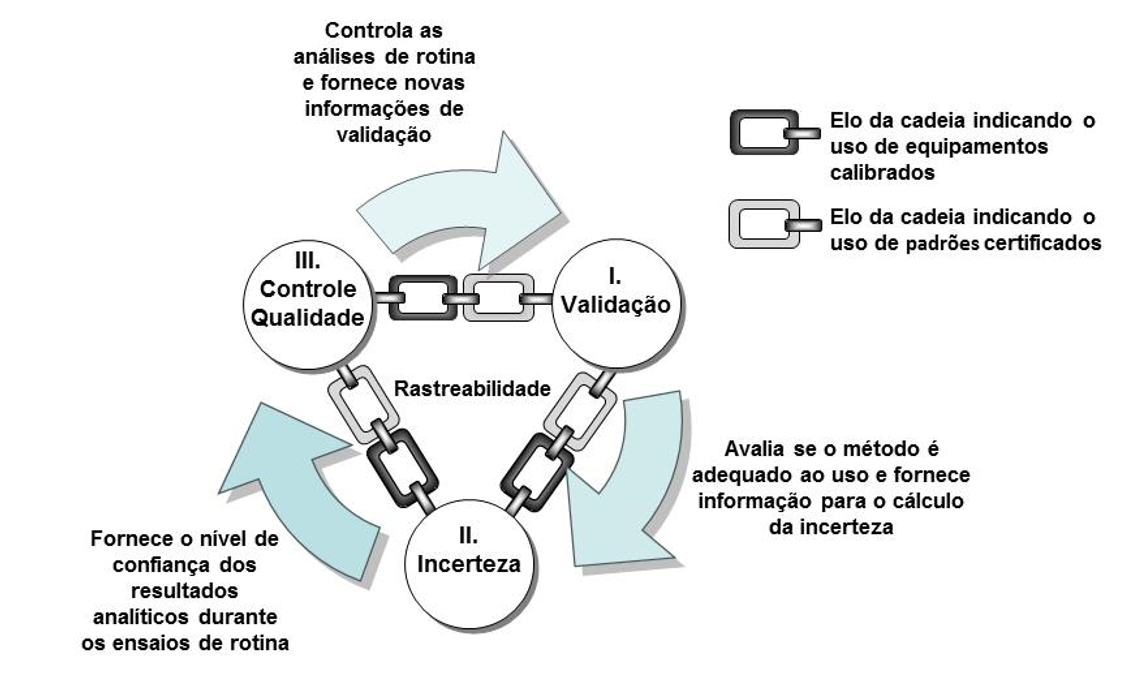
A relação entre validação, incerteza e controle de qualidade pode ser entendida ao implantar um novo método de ensaio em um laboratório.
Inicialmente, é necessário validar o método visando demonstrar que é adequado ao uso pretendido, destacando que, durante este processo, será possível conhecer os pontos fortes e fracos do método avaliado. Mesmo que o método seja normalizado, sempre é necessário que se avalie algum parâmetro de validação para verificar se o laboratório consegue obter resultados adequados com o método normalizado em suas condições operacionais (funcionários, instalações e equipamentos) assim, para implantar um novo método de ensaio em rotina, é necessário aplicar o primeiro requisito do AQAC, a validação.
Após a validação, o segundo passo leva à estimativa da incerteza de medição, lembrando que aplicando a abordagem Validation Based é possível utilizar de alguns resultados da validação, como “entradas” no diagrama de Ishikawa para o cálculo de incerteza (como, por exemplo, o estudo de linearidade e repetibilidade). Desta maneira, poderá ser determinado o nível de confiança que se tem no resultado conhecendo o valor da incerteza (aplicando, assim, o segundo requisito do AQAC).
Após avaliar o método (durante a validação), ter o nível de confiança do resultado (conhecendo a incerteza), o terceiro e último passo do ciclo será a aplicação do controle de qualidade, que visa demonstrar, durante cada batelada de ensaio, que o método ainda pode proporcionar resultados confiáveis, em outras palavras, é um processo de validação constante.
Considere que, para cada batelada de amostra, seja realizada a análise de uma amostra de concentração conhecida, assim, é possível avaliar, de imediato, a exatidão e, a longo prazo, calcular a dispersão de uma série de resultados, obtendo-se, assim, uma precisão intermediária. Produzindo novos dados de precisão intermediária (que é um parâmetro de validação), é possível recalcular a incerteza, assim o AQAC acaba girando como um círculo sem fim. Destaca-se que, para dar sustentabilidade à aplicação destes requisitos, o ciclo é ligado por elos de uma corrente indicados como “uso de equipamentos calibrados” e “uso de padrões certificados”, necessários para a confiabilidade dos resultados obtidos.
Considerando os requisitos básicos para prover confiabilidade e rastreabilidade de resultados, o AQAC pode ser aplicado como uma poderosa ferramenta da qualidade para qualquer tipo de laboratório. Para laboratórios que apresentam um sistema de qualidade formal, como, por exemplo, ISO/IEC 17025 ou BPL (que já aborda alguns requisitos do AQAC), sua aplicação resultará no suporte e melhoria contínua da qualidade dos resultados. No entanto, o AQAC também fornece suporte para rastreabilidade, confiabilidade e qualidade dos resultados para um laboratório que não tem um sistema formal de qualidade, como, por exemplo, laboratórios de pesquisa, podendo ser, talvez, uma futura ferramenta para avaliação ou, quem sabe, até acreditação para este tipo de laboratório [01].
Referências[01] OLIVARES, Igor Renato Bertoni. Gestão de Qualidade em Laboratórios (3a edição – revisada e ampliada). 3. ed. Campinas: Átomo e Alínea, 2015. 160 p (3ª edição em Português e Inglês).

 VALIDAÇÃO, INCERTEZA E
CONTROLE DE QUALIDADE
VALIDAÇÃO, INCERTEZA E
CONTROLE DE QUALIDADE



